The Pitt Hopkins Research Foundation is committed to directly funding the most promising research available in the world to help find a treatment and ultimately a cure for Pitt Hopkins syndrome.
We do more than just hold fundraisers and write checks. We are defining the landscape of Pitt Hopkins research. We set the research strategy by proactively identifying and monitoring promising therapeutic areas, seeking out scientific and industry partnerships and working closely with them to advance programs through the drug development pipeline. We believe Pitt Hopkins can be cured, but time matters. A rigorous, coordinated, and agile research agenda will get us there as quickly as possible.
Our Progress: Since our beginning in 2012, we have funded over $6,000,000 in research grants and awards. After only eight years (a record timeline for scientific discovery) we launched our first clinical trial for Microbiota Transfer Therapy with Arizona State University in 2019. Since then we’ve launched two more clinical trials and are preparing to launch another two within the next 12 months (April, 2025). All the while, we are working aggressively to bring more drugs and more therapeutics to trial and are actively pursuing various genetic therapies.
GENE THERAPY
We believe genetic therapies are our best chance at meaningful treatments and we are heavily pursuing those with more than 6 labs devoted to various approaches. We are taking every shot we can to make our dreams of meaningful gene therapy a reality.
Our efforts have attracted attention from university and pharmaceutical scientists, the National Institute of Mental Health (NIMH), and the California Institute for Regenerative Medicine (CIRM). The NIMH and the CIRM have supported our seed grants with $7,619,000 in additional research dollars.
We are the only organization in the world devoted to funding research for PTHS.
Our Future:
Our research focus areas for 2025 include:
- Bring our first gene therapy to trial in partnership with Mahzi Therapeutics
- Complete our Phase 3 Microbiota Transfer Trial (MTT) in partnership with ASU
- Launch a clinical trial to determine the efficacy of Vorinostat in PTHS in partnership with Unravel Biosciences
- Continue to transition from basic research to transformational, applied discoveries
- Fund additional clinicians and clinical researchers
- Fund small molecule drug discovery program targeting the TCF4 pathway
- Fund and broaden our gene therapy programs
- Fund mRNA research
- Fund artificial intelligence platforms to search for possible existing therapeutics
- Collaborate with private pharmaceutical companies to assist in clinical trials
- Fund the study of clinical phenotyping, outcome measures and potential biomarkers
Research into these key programs requires funding. WE NEED YOUR HELP! Your tax-deductible gift will give so much more than money – it gives all of our families HOPE! Our ultimate goal is to close our doors for good… by finding a cure!
This research has far reaching effects into the science of memory and learning. Because we already KNOW the gene that causes PTHS, TCF4, we are in an important place to fund meaningful research, research that could possibly give insight to other learning, memory and motor disorders with no known cause, like Autism, Alzheimer’s, Epilepsy and Parkinson’s.
OVER 94% OF ALL FUNDS DONATED GO TO RESEARCH
Progress from our Labs:
Pushing the Boundaries in Genetic Therapy
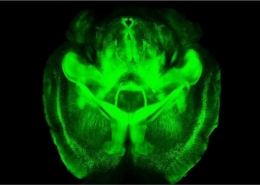
Milestone Achieved: U7 snRNA Therapy Boosts TCF4 in Mouse Model
The Research Institute at Nationwide Children’s Hospital
We’re excited to share a major research milestone in our efforts to develop gene therapies for Pitt Hopkins syndrome. Initiated by Dr. Kathrin Meyer’s lab at Nationwide Children’s Hospital and now being advanced by Dr. Afrooz Rashnonejad at the same institution, this project focused on using a novel approach—repurposing U7 small nuclear RNA (snRNA)—to modulate TCF4 transcript expression.
In the first phase of this study, the team successfully demonstrated increased TCF4 protein expression in a mouse model of PTHS, marking an encouraging step forward. This innovative strategy holds strong potential for addressing the underlying genetic cause of the syndrome. As we move into the next phase, our researchers will continue to refine the approach in the hopes of bringing it to clinical trials.
CRISPR Progress: Restoring TCF4 with a Smaller, Smarter Tool
Fink Lab at UC Davis
We’re proud to share exciting progress from Dr. Kyle Fink’s lab at UC Davis, where a cutting-edge gene activation strategy is showing real promise for Pitt Hopkins syndrome (PTHS). PTHS is caused by changes in a gene called TCF4, which plays a key role in brain development and function. Right now, there are no treatments that address the root cause of PTHS—but that’s what this research aims to change.
With previous funding from the Pitt Hopkins Research Foundation, Dr. Fink’s team showed they could successfully boost TCF4 levels in cells using a tool called CRISPR activation. This system was effective, but the version they used was too large to fit into the delivery system (AAV) typically used in gene therapy.
Now, with continued support, the Fink lab is taking the next big step: testing a new, much smaller CRISPR system called casMINI. This compact version can fit into a single AAV, making it far more practical for future therapies. The team is currently screening the best guide combinations to help restore TCF4 expression—and we’re hopeful this could pave the way for a powerful, precise treatment for PTHS.
Small RNA Activation Therapy for Pitt Hopkins
Corey Lab at UT Southwestern
The root cause of Pitt Hopkins syndrome is insufficient production of a critical protein called TCF4. Correcting this deficit at the genetic level remains one of the most promising paths toward a meaningful treatment.
Dr. Corey’s lab is pioneering the use of small activating RNAs (saRNAs)—short RNA molecules designed to precisely target and “turn on” genes that aren’t making enough protein. In this case, the therapy will focus on increasing expression of TCF4 by binding to regulatory regions of the gene, effectively boosting its activity.
The Corey Lab has spent years refining this approach and will now test its potential in Pitt Hopkins. Their immediate goal is to determine whether sbRNA can reliably elevate TCF4 protein levels—a critical proof-of-concept milestone expected within six months.
This work is part of the Foundation’s larger strategy to invest in multiple, complementary approaches to increase TCF4 and drive progress toward a cure.
We are honored to support this cutting-edge work and grateful to Dr. Corey and his team for their dedication to our children.
Read more about the Corey Lab’s work at coreyutswlab.com
In our talks with David Corey and his collaborator Bethany Jankowski, it’s clear they are most concerned with discovering a viable treatment for our children as soon as possible. And to us, that is the epitome of Being Better. Thank you Victor, for giving us the inspiration and fuel to continue fighting. Read more about this Victor Pauca Be Better Award here.
The Research Rundown: 10 Key Updates You Need to Know
1. Three Clinical Trials and Counting
Since 2023, we’ve launched three clinical trials: one with Neuren Pharmaceuticals targeting BDNF enhancement, and two at Arizona State University exploring Microbiota Transfer Therapy.
2. Gene Therapy is Coming
Mahzi Therapeutics has licensed our UCSD gene therapy work and is on track to file an IND this summer, with a first-in-human trial planned for late 2025 or early 2026.
3. Repurposing Old Drugs for New Hope
Unravel Therapeutics is preparing a clinical study in Medellín, Colombia using a repurposed drug for Pitt Hopkins syndrome, supported by Colombia’s fast-track orphan disease program.
Click here to see our full therapeutic pipeline.
4. Expanding Our Clinical Network
We are growing a powerful network of clinical partners. The Neuren trial included five sites—Colorado Children’s, UAB, Chicago Rush, and more—while Mahzi has already begun identifying its own trial sites.
5. Building Smarter with Data
To prepare for Mahzi’s trial (as well as other future trials), we’ve partnered with Citizen Health to collect and analyze medical records. So far, ~60 families have contributed to this crucial data initiative.
6. Join Our Registry
Our patient registry with Sanford is growing steadily. If you haven’t already, please join—it’s one of the most important ways to accelerate research and access future trials.
7. Biomarker Discovery is Underway
We recently collected over 30 blood and stool samples for analysis by Metabolon, a leader in metabolomics. The study is complete and now under review.
8. New Mouse Models in Development
Beyond our original heterozygous knockout mice, two new models are now in development: a humanized model at Jackson Labs and a second from the Philpot Lab.
9. Exploring ASO Therapy
We’ve been funding early ASO (antisense oligonucleotide) development at La Jolla Labs. Initial data is promising, and we’ll soon begin testing in organoids at the Muotri Lab.
10. Every Step Brings Us Closer
Each of these efforts—clinical trials, gene therapy, patient data, biomarkers, and new models—moves us closer to effective treatments and, ultimately, a cure.
From Lab to Clinic, We are Transforming Lives!
Pioneering Progress: Driving Clinical Trials with Unyielding Passion!
At the PHRF, we don’t just fund research, we fund strategic, cultivated projects that lead to viable and timely treatments in the clinic. Our commitment to launching clinical trials has never wavered, and in 2021, just 8 years after we started the foundation we launched our first-ever clinical trial: Microbiota Material Transfer Therapy. Read more about it below:
Groundbreaking Clinical Trial Advances: FDA Review Underway for Revolutionary Treatment
Microbiota Material Transfer Trial – PI James Adams and Rosy Khajelmik;Arizona State University
In 2021 The Pitt Hopkins Research Foundation launched its first ever clinical trial: Microbiiota Transfer Therapy. The trial finished in 2022 with great success in reducing the severe and debilitating gut issues our children suffer with daily. As of April 2023, this treament is being reviewed by the FDA for possible approval. The fact that we have a succesful treatment to bring to the FDA for approval is unprecedented for such a young and small foundation
In 2024 we launched a Phase @ MMT trial, this time using a powder formulation that we hope will make the treatment more accessible for those who cannot swallow pills.
Neuren NNZ-2591
Neuren Pharmaceuticals is the innovative company behind Daybue, the first treatment ever approved for Rett Syndrome. When they needed another population to trial their next compound, NNZ-2591 they called on The Pitt Hopkins Research Foundation because of our strong ties to our committed community.
Their drug NNZ-2591 has shown strong efficacy in a mouse model of Pitt Hopkins syndrome and been granted Orphan Drug Designation by both the US FDA and the European Drug Regulator, the EMA. Pre-clinical studies with NNZ-2591 have shown that it appears to normalize the underlying biology of the brain, and suggest that NNZ-2591 may have a favorable effect on the broad features of Pitt Hopkins syndrome, rather than being restricted to treating an isolated symptom or symptoms.
The purpose of the study is to look at the safety, tolerability, and efficacy of NNZ-2591 in the treatment of children with Pitt Hopkins syndrome.
UP NEXT…
Gene Therapy Breakthrough: Unlocking the Future of Healing
Our next focus, in conjunction with Mahzi Therapeutics, is bringing our first-ever gene therapy to trial. The therapy concept was proven in the lab of Dr. Allyson Muotri at UCSD. It was then successfully trialed in newborn mice in the Lab of Dr. Patricia Cogram at the University of Chile. Currently, it is being tested for toxicity in primates in efficacy in older mice.
Apply for Grants
Our goal is to fund research directly targeted toward finding therapeutic treatments for Pitt Hopkins Syndrome.
Proposals should be no more than five pages and targeted towards 1 year of funding with a maximum award amount of 75K.
Proposals are accepted on a rolling basis. For questions and specific proposal guidelines please email audrey.davidow@gmail.com
All grant recipients, as part of the award contract, will be asked to hold quarterly conference calls with each other in order to promote and encourage real time communication and expedite the discovery process.
The PHRF does not pay any indirect costs associated with grant funding.
OVER 94% OF ALL FUNDS DONATED GO TO RESEARCH
Our board and officers are parents and professionals who work diligently to ensure over 94% of all funds donated go toward research and our mission of finding a cure.